✨"I am among those who think that science has great beauty. A scientist in his laboratory is not only a technician; he is also a child placed before natural phenomena which impress him like a fairy tale." - Marie Curie
✨"Be less curious about people, and more curious about ideas." - Marie Curie
✊🏽 Marie Curie - Polish & naturalized French physicist & chemist who conducted pioneering research on radioactivity.
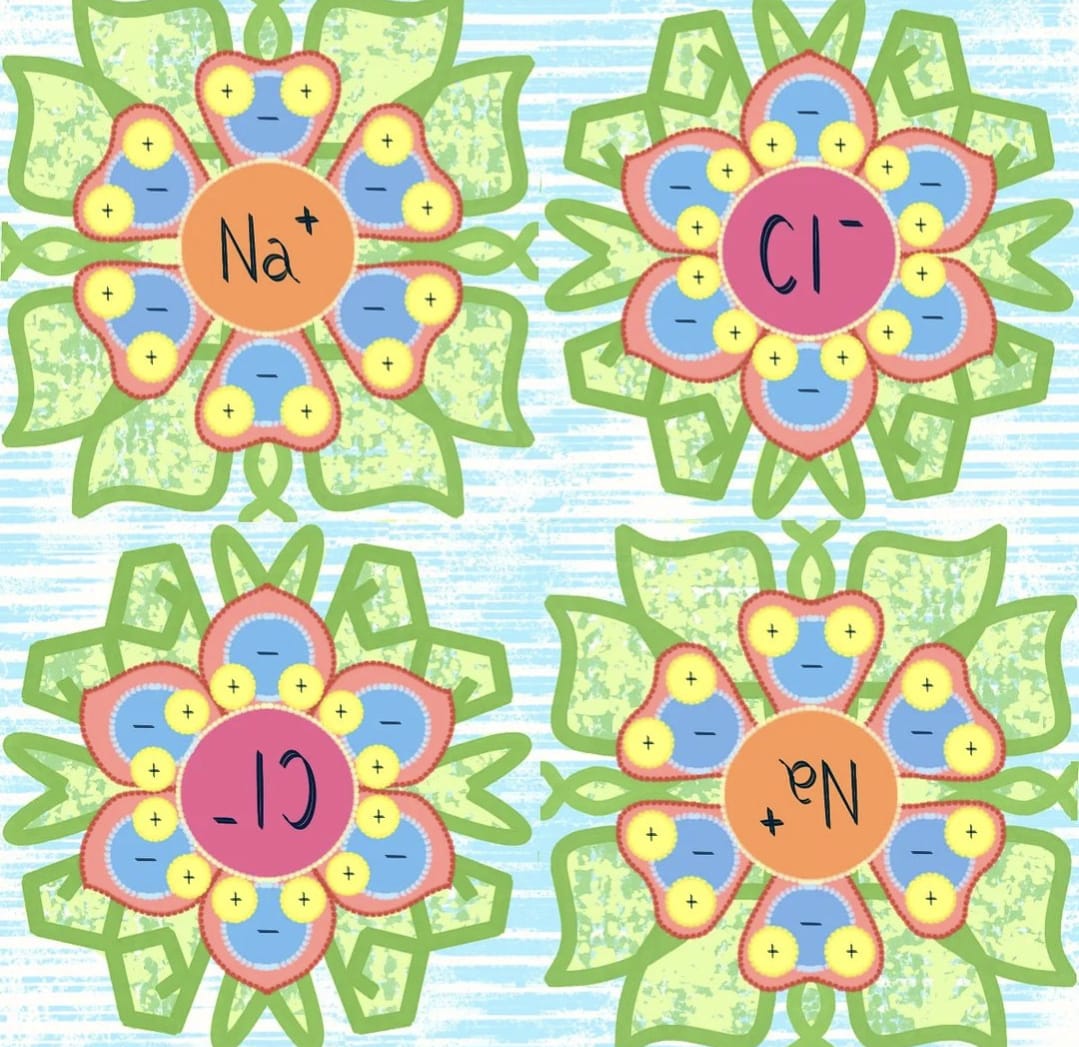
🌼 Day 19: OPPOSITES - Chemical Bonds, Ionic Bonding, NaCl = Sodium chloride aka Table Salt
Digital Art
👩🏽🔬 A chemical bond is a lasting attraction between atoms, ions, or molecules that enables formation of chemical compounds. The bond may result from the electrostatic force of attraction between oppositely charged ions as in ionic bonds or through the sharing of electrons as in covalent bonds.
👩🏽🔬 Ionic bonding is a type of bond that involves an electrostatic attraction between OPPOSITELY charged ions, typically occurring between a metal & non-metal. This bond is formed when valence electrons (outer shell) of one atom are transferred permanently over to another atom. The atom that loses the electron becomes positively charged (cation) while the one that gains it becomes negatively charged (anion).
👩🏽🔬 NaCl is an ionic chemical compound because the bond formed is due to the transfer of electrons from Sodium atom (Na) to Chlorine atom (Cl), which results in the formation of a Sodium ion Na+ (cation) and a Chloride ion Cl– (anion). These ions are attracted by the electrostatic force of attraction and forms the ionic bond resulting in the formation of Sodium chloride aka table salt.
👩🔬 The Bohr & polarity diagrams of these atoms looked like flowers to me, so I played off of them even more. 😜